Reduce global product label generation time from months to days with an easy and cost-effective system your whole team will love.

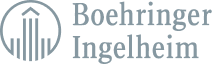


ComplianceAuthor® AI simplifies your internal processes around authoring, reusing, translating, and managing product labels.
ComplianceAuthor® AI uses natural language processing specifically designed for pharmaceutical companies to index your existing clinical information into one controlled system.
ComplianceAuthor AI allows users easy and fast authoring through MS-Word. This provides companies with a powerful xml system in a simple Word interface.
ComplianceAuthor® AI helps users, health authorities and authorized third parties easily collaborate. Harmonize internal and external comments, edits, and tracked changes automatically in one document.
ComplianceAuthor® AI manages technical translations out of the box. ComplianceAuthor AI supports both in-house and third-party translation service providers.
ComplianceAuthor® AI leverages state of the art omni-channel publishing capabilities to automatically generate regulatory submissions like QRD, SPL, and FHIR formats.
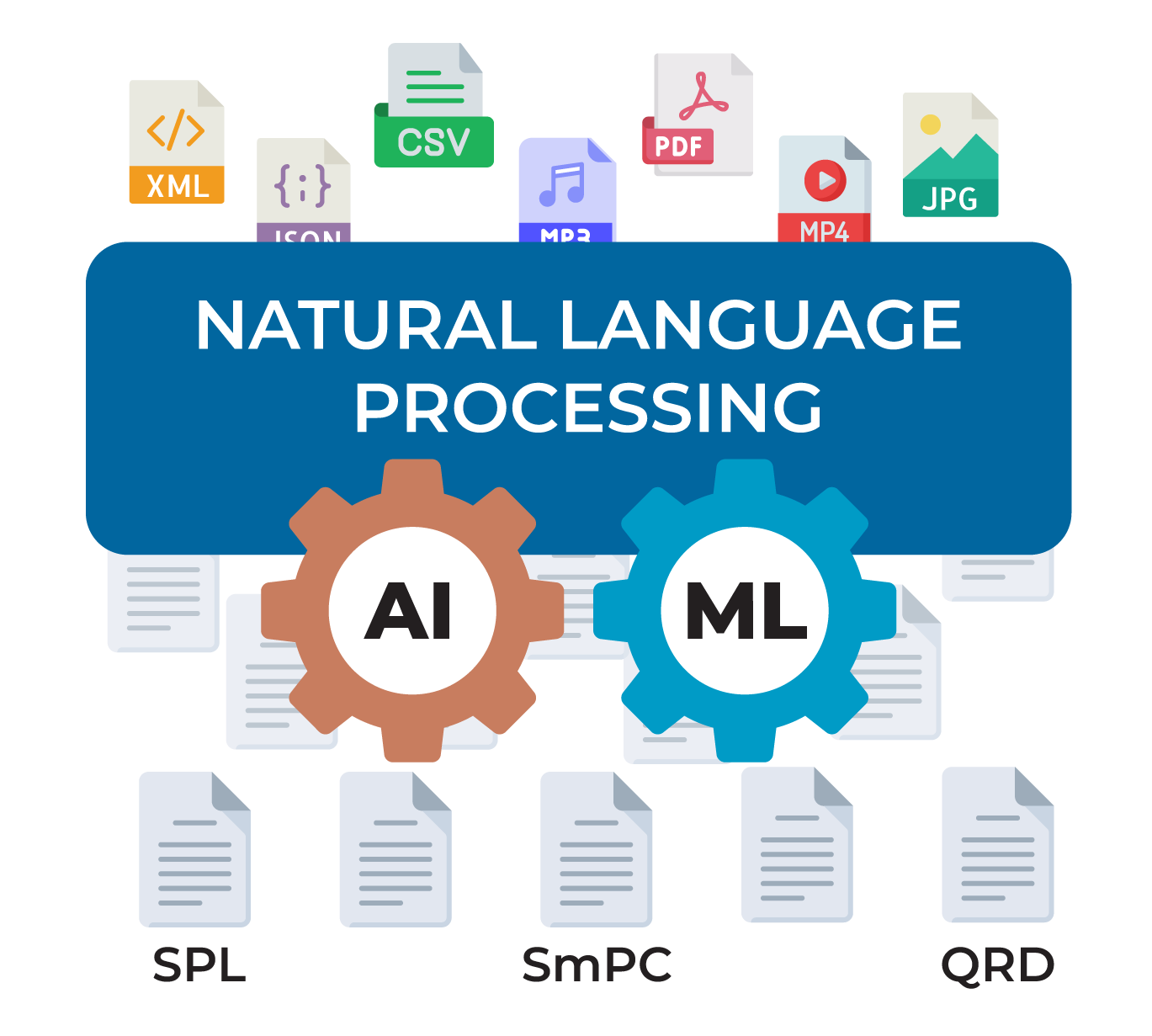
Create and deploy structured content components across the globe. Make a change once, and gain insights to determine the next best action.
Experience seamless compatibility with all health authority outputs, ensuring your labels are ready for every market.
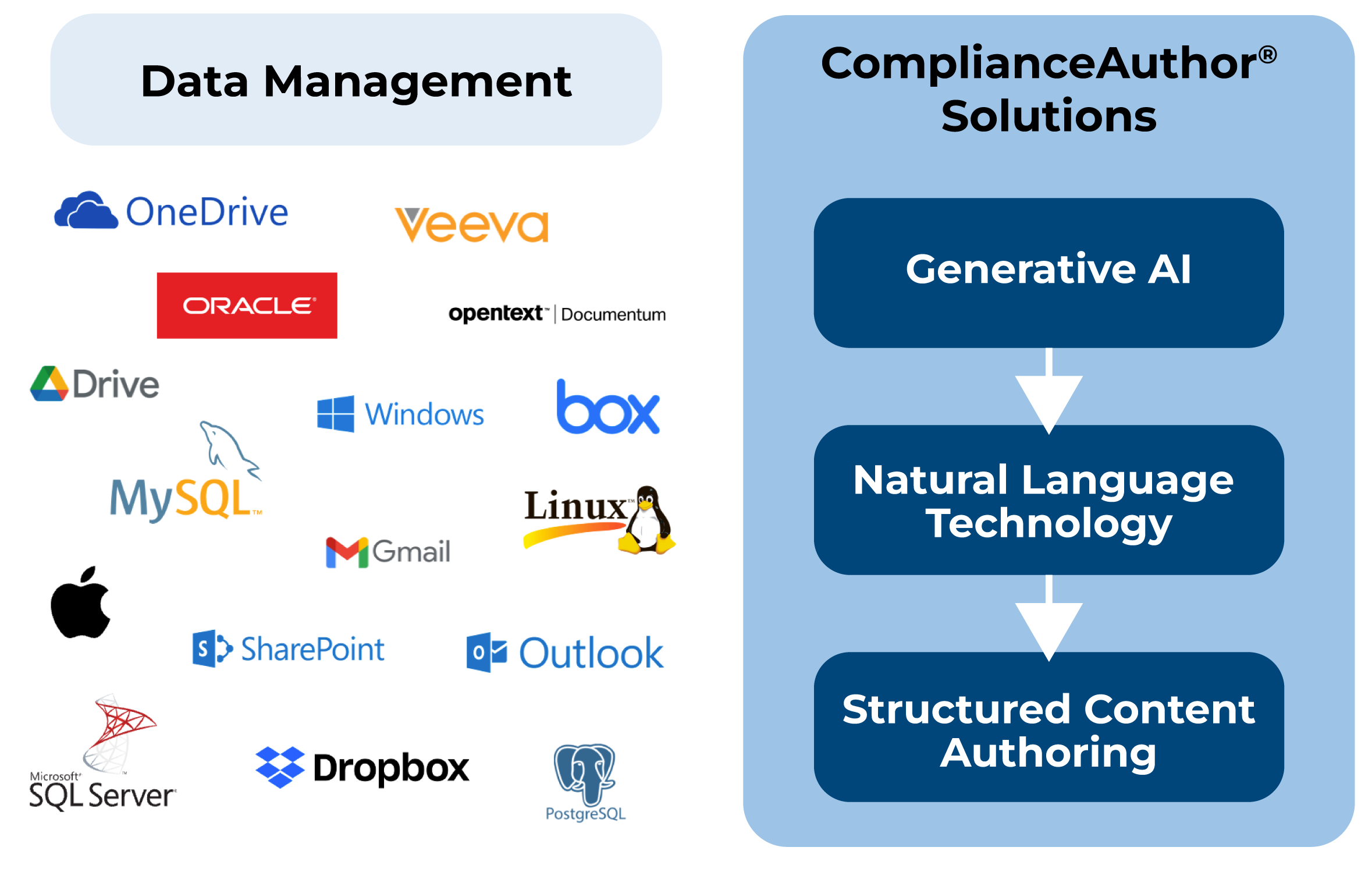
ComplianceAuthor® AI integrates easily with your current systems in a way amplifies your capabilities.
Search, find and index and monitor content. Manage change control across your enterprise, leveraging machine learning.
Design your product labels with regulated and compliant components at every stage of the drug’s lifecycle.
Use generative AI to transform internal data sheets into approved health authority labels, with a human in the loop.